| Pure Water Products |
|
March, 2026
|
|
|
Water Treatment Issues and Current Water News |
In this early Spring Occasional, you'll hear about the Pure Water Gazette's giant trove of water information, hear about chlorine burns from Gazette technical wizard Pure Water Annie, learn a lot about iron in well water, as explained by the Minnesota Dept. of Health,find out about the courts' banning of the first Trump administration's ban on the previous administration's ban on Dursban, the best home water treatment for PFAS, quartz sleeve maintenance as explained by Mike, the alarming, faster-than-expected rise in sea level, new creatures from the oceans, the probably doomed-to-fail effort to alter the ocean's pH chemically, SIX (aka suspended ion exchange), a tree in India that emits Holy Water, the mysterious absence of fluoride in Birmingham's public water, plastic water bottles, the "Drought Paradox" in the Colorado River Basin, important new evidence to explain water's strange behavior, and, as always there is much, much more.
|
Water and Environmental Issues |
More from the Pure Water Gazette Bulging Article Archive
The Pure Water Gazette website has been archiving information about water treatment for about three decades. Since we add regularly and almost never discard, the Gazette website is now bulging with many hundreds of informative and useful articles about water issues and how to deal with them. Best of all, there are no intrusive pop-ups or other distractions from the subject at hand. Even more importantly, information-rich articles don't disappear. You can depend on them to be there for future reference.
The Pure Water Gazette is a non-commercial site. Its function is to provide information, not sell products. The Gazette website is designed to be useful and informative, not beautiful. It is arranged into several broad categories. Below are the main topics from the site's sidebar index. Each of the main categories leads to articles added to the site since 2012.
-
- A few clicks on the links above will give an indication of the extensiveness of the Gazette's content. In this issue of the Occasional we're going to include some miscellaneous articles. One illustrates the importance of archiving. The excellent article about iron treatment from the Minnesota Health Department has been taken down from the state's website and would be lost had we not made a copy. Gazette water consultant Pure Water Annie's explanation of chlorine burns explains the need for this unpopular event.
What Are Chlorine Burns?
by Pure Water Annie
Gazette technical wizard Pure Water Annie addresses the perplexing questions about water treatment.
Once a year, usually in spring, water suppliers that normally disinfect their product with chloramine, a mixture of chlorine and ammonia, perform a cleaning procedure known as a “chlorine burn.” The purpose is simply to clean out the pipes, ridding the distribution system of film and debris that has built up.
The clean-out is accomplished by simply switching disinfectants from chloramine to straight chlorine for a time, and usually upping the dosage a bit to speed things along. Compared with chlorine, chloramine is a rather weak disinfectant. Its weak performance allows sludge and scum, bacterial film, to build up in pipe walls and crevices. The yearly purge, or “burn,” with straight chlorine cleans things out.
Chloramine is substituted for chlorine as the regular disinfectant in an increasing number of city water systems. The switch from chlorine to chloramine has been going on over a number of years as suppliers seek ways to stay in compliance with EPA standards for DBPs, disinfection by-products, that are produced as a consequence of chlorination. Some DBPs are known carcinogens, and EPA requires suppliers to monitor them. Chloramine, a weaker disinfectant, does not produce DBPs.
Are chlorine burns a good idea? Good or bad, they are necessary, since without a periodic cleanout, buildup in pipes would create significant problems for the water system. The practice does call into question, however, the wisdom of using chloramine rather than chlorine in the first place, since, as many argue, the burn and subsequent purging of pipes creates elevated levels of disinfection by-products in the system and higher than normal chlorine discharge into lakes and streams. In other words, for a short time we get concentrated doses of disinfectants and byproducts, which may be worse than what we would have with chlorine as the regular disinfectant.
The moral: With a good carbon filtration system in your home, you won’t even know when the burn takes place. The elevated chlorine levels, murky water, and dislodged sediment that your neighbors are complaining about, you won’t even notice. If your city uses chloramine, you should use equipment that is designed for chloramine treatment. Any filter that removes chloramine also removes the chlorine used during the annual burn.
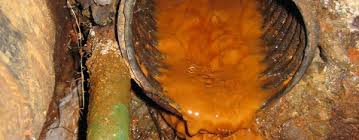
Iron in Well Water
Gazette Introductory Note: Iron is among the most persistent problems faced by residential well owners. It is also among the least understood. The following article from the Minnesota Department of Health website is a concise overview of the iron issue. Our main website, www.purewaterproducts.com, has in-depth information on the treatment of iron by a variety of methods. We also welcome phone or email information requests about treatment of iron issues in residential wells.–Gene Franks, Pure Water Products.
What do these have in common – a taconite mine in northern Minnesota, the color of your blood, a rusty pail, and yellow or red stains on sinks and plumbing fixtures? The answer is – Iron. Iron is the fourth most abundant mineral in the earth’s crust. Soils and rocks in Minnesota may contain minerals very high in iron, so high in fact, that taconite can be mined for its iron content. Iron gives the hemoglobin of blood it’s red color and allows the blood to carry oxygen. The iron in a metal pail turns to rust when exposed to water and oxygen. In a similar way, iron minerals in water turn to rust and stain plumbing fixtures and laundry.
Iron in Well Water
As rain falls or snow melts on the land surface, and water seeps through iron-bearing soil and rock, iron can be dissolved into the water. In some cases, iron can also result from corrosion of iron or steel well casing or water pipes.
Health Concerns
Iron in well water usually does not present a health problem. In fact, iron is needed to transport oxygen in the blood. The human body requires approximately 1 to 3 additional milligrams of iron per day (mg/day). The average intake of iron is approximately 16 mg/day, virtually all from food such as green leafy vegetables, red meat, and iron-fortified cereals. The amount of iron in water is usually low, and the chemical form of the iron found in water is not readily absorbed by the body. Iron bacteria, that may be associated with iron in water, are not a health problem.
Iron may present some concern if certain bacteria have entered a well, since some pathogenic (harmful) organisms require iron to grow, and the presence of iron particles makes elimination of the bacteria more difficult.
Iron Problems
Iron Stains in Tub
Iron in water can cause yellow, red, or brown stains on laundry, dishes, and plumbing fixtures such as sinks. In addition, iron can clog wells, pumps, sprinklers, and other devices such as dishwashers, which can lead to costly repairs. Iron gives a metallic taste to water, and can affect foods and beverages – turning tea, coffee, and potatoes black.
Forms of Iron
Iron can occur in water in a number of different forms. The type of iron present is important when considering water treatment. Water that comes out of the faucet clear, but turns red or brown after standing is “ferrous” iron, commonly referred to as “ clear-water” iron. Water which is red or yellow when first drawn is “ferric” iron, often referred to as “ red- water” iron. Iron can form compounds with naturally occurring acids, and exist as “ organic” iron. Organic iron is usually yellow or brown, but may be colorless. Water containing iron bacteria is said to contain “ bacterial” iron. Testing
Yellow or red colored water is often a good indication that iron is present. However, a testing laboratory can determine the exact amount of iron, which can be useful in determining the best type of treatment. In addition to testing for iron, it can be of value to also test for hardness, pH, alkalinity, and iron bacteria. County health departments may offer some of these tests. Private testing laboratories can be contacted about their services and fees. Most advertise in the phone book under “Laboratories-Testing.”
The amount of a dissolved material in water is usually reported as the number of milligrams per liter (mg/L). This is the weight of material in 1 liter (approximately 1 quart) of water. A milligram per liter is approximately equal to 1 part per million (ppm). Iron in amounts above 0.3 mg/L is usually considered objectionable. Iron levels are usually less than 10 mg/L.
Controlling Iron
The most common method for controlling iron in water is water treatment. In some circumstances, another alternative is to use a different water source that is low in iron, such as a public water system or a well drawing water from a different water-bearing formation. In some cases, a new well may be an option, however, it is difficult to predict what the iron concentration will be. Neighboring wells may be an indicator, but the iron content of two nearby wells may be quite different.
Water Treatment
Treatment of water containing iron depends on the form(s) of the iron present, the chemistry of the water, and the type of well and water system.
Clear-water iron is most commonly removed with a water softener. Manufacturers report that some units are capable of removing up to 10 mg/L, however 2 to 5 mg/L is a more common limit. A water softener is actually designed to remove hardness minerals like calcium and magnesium. Iron will plug the softener, and must be periodically removed from the softener resin by backwashing. Also, if the water hardness is low and the iron content high, or if the water system allows contact with air, such as occurs in an air-charged “galvanized” pressure tank, a softener will not work well. Ion exchange water softeners add sodium to the water which may be a concern for persons on a sodium restricted diet.
Red-water iron can be removed in small quantities by a sediment filter, carbon filter, or water softener, but the treatment system will very quickly plug up. A more common treatment for red-water iron and clear-water iron in concentrations up to 10 or 15 mg/L is a manganese greensand filter, often referred to as an “iron filter.” Aeration (injecting air) or chemical oxidation (usually adding chlorine in the form of calcium or sodium hypochlorite) followed by filtration are options if iron levels exceed 10 mg/L.
Organic iron and tannins present special water treatment challenges.Tannins are natural organics produced by vegetation which stain water a tea-color. In fact, the tannins in coffee or tea produce the brown color. When tea or coffee is made with water containing iron, the tannins react with the iron forming a black residue. Organic iron is a compound formed from an organic acid and iron. Organic iron and tannins can occur in very shallow wells, or wells being affected by surface water. Organic iron and tannins can slow or prevent iron oxidation, so water softeners, aeration systems, and iron filters may not work well. Chemical oxidation followed by filtration may be an option.
Well Treatment
Iron bacteria are organisms that consume iron to survive and, in the process, produce deposits of iron, and a red or brown slime called a “biofilm.” The organisms are not harmful to humans, but can make an iron problem much worse. The organisms naturally occur in shallow soils and groundwater, and they may be introduced into a well or water system when it is constructed or repaired.
Treatment options for elimination or reduction of iron bacteria include physical removal, heat, and chemical treatment. The most common treatment is “shock” chlorination of the well and water system. See Iron Bacteria in Well Water. Remember, iron bacteria need iron to survive. Eliminating the bacteria will not eliminate the iron – both well treatment for the bacteria, and water treatment for the iron will be needed.
Trump Reversal of Chlorpyrifos Ban Reversed by Court
A federal appeals court ruled August 9 that the Trump administration endangered public health by keeping a widely used pesticide on the market despite extensive scientific evidence that even tiny levels of exposure can harm babies’ brains.
The 9th U.S. Circuit Court of Appeals in San Francisco ordered the Environmental Protection Agency to remove chlorpyrifos from sale in the United States within 60 days.
A coalition of farmworkers and environmental groups sued last year after then-EPA chief Scott Pruitt reversed an Obama-era effort to ban chlorpyrifos, which is widely sprayed on citrus fruit, apples and other crops. The attorneys general for several states joined the case against EPA, including California, New York and Massachusetts.
Chlorpyrifos was created by Dow Chemical Co. in the 1960s. It remains among the most widely used agricultural pesticides in the United States, with the chemical giant selling about 5 million pounds domestically each year.
Chlorpyrifos is marketed under such brand names as Dursban and Lorsban
Chlorpyrifos belongs to a family of organophosphate pesticides that are chemically similar to a nerve gas developed by Nazi Germany before World War II. As a result of its wide use as a pesticide over the past four decades, traces of chlorpyrifos are commonly found in sources of drinking water. A 2012 study at the University of California at Berkeley found that 87 percent of umbilical-cord blood samples tested from newborn babies contained detectable levels of the pesticide.
In October 2015, the Obama administration proposed banning the pesticide’s use on food. A risk assessment memo issued by nine EPA scientists concluded: “There is a breadth of information available on the potential adverse neurodevelopmental effects in infants and children as a result of prenatal exposure to chlorpyrifos.”
Federal law requires EPA to ensure that pesticides used on food in the United States are safe for human consumption — especially children, who are typically far more sensitive to the negative effects of poisons.
Shortly after his appointment by President Donald Trump in 2017, Pruitt announced he was reversing the Obama administration effort to ban chlorpyrifos, adopting Dow’s position that the science showing chlorpyrifos is harmful was inconclusive and flawed.
The Associated Press reported in June 2017 that Pruitt announced his agency’s reversal on chlorpyrifos just 20 days after his official schedule showed a meeting with Dow CEO Andrew Liveris. At the time, Liveris headed a White House manufacturing working group, and his company had written a $1 million check to help underwrite Trump’s inaugural festivities.
Pruitt resigned July 6 amid more than a dozen ethics investigations.
Little information is available about point of use treatment of chlorpyrifos in water, but treatment with GAC has been used with some success in wastewater treatment. Chlorpyrifos has also been reduced with ozone and coagulation. Some states now have established standards for chlorpyrifos. Florida, for example, has a legal maximum of only 21 parts per billion.
Home Treatment Strategies for PFAS
Either Reverse Osmosis or an Undersink Filter Can Provide PFAS-free Drinking Water
A high quality undersink filter with lots of carbon provides excellent protection against the “forever chemicals” PFAS in drinking water.
For drinking water, reverse osmosis is a sure thing. It removes PFAS handily. A good carbon drinking water filter is also a reliable PFAS reducer. High quality carbon block cartridges have an impressive range for PFAS. Our standard double undersink filter with MatriKX PB1 and CTO+ cartridges is rated for over 6000 gallons of PFAS removal between cartridge changes. That’s a lot of drinking water. The usual “change once a year” recommendation applies.
For point of entry (whole house) treatment, if you want high PFAS reduction you’ll need to reduce your service flow rate and change cartridges often. A single MatriKX CTO+ in whole house 4.5″ X 20″ size has a 21,000 gallon PFAS reduction capacity at 3 gallons per minute. Two of these installed in parallel would give a 42,000 gallons capacity at a service flow of six gallons per minute.
PFAS can also be effectively treated with ion exchange. This strategy is often used by municipal water suppliers, but is not currently as practical or as affordable for home use as carbon filtration and reverse osmosis.
Quartz Sleeve: Essential to UV Performance
Maintaining the Quartz Sleeve

Certified Viqua Installer Mike aims a disapproving eye at a customer’s quartz sleeve during a service call in Flower Mound, TX
An essential part of any ultraviolet water purification system is a clear tube that looks like glass but is really made of quartz. It forms a barrier between the UV lamp and the water being treated. The tube is called a quartz sleeve. The UV lamp is housed inside the sleeve and the water is on the outside, so the lamp’s UV dosage that actually gets to the water depends on how clean and clear the quartz sleeve is.
Ultraviolet units are normally preceded by a five micron sediment filter to assure that the water being treated does not contain particles large enough to provide shade that would protect microbes from the germicidal lamp. The filter, however, does not remove minerals in the water that can form scale on the outside of the very hot quartz sleeve. The most common scaling agents are hardness (calcium and magnesium) and iron.
Regular maintenance of a UV system includes examining, cleaning, and, if necessary, replacing the quartz sleeve to assure maximum UV transmittance. Recommended cleaning is with a clean, lint-free cloth soaked in vinegar or another mild acid. It is also highly recommended that you handle with care, don’t leave finger prints, and don’t break the sleeve. Especially don’t break it.
The sleeve cleaned up nicely with white vinegar and some scrubbing. Mike is happy.
Manufacturers’ quartz sleeve recommendations vary, some recommending sleeve change with every second lamp change even if the sleeve appears clear to the eye. This is because UV light can degrade the quartz and block efficient UV transmittance even if the sleeve appears to be crystal clear.
|
Water News
Water News for March of 2026

Water News For March 2026
Sea Levels Are Rising Faster than Predicted
Sea levels around the world have been underestimated due to inaccurate modelling, with research suggesting ocean levels are far higher than previously understood. The finding could significantly affect assessments of the future impacts of global heating and the effects on coastal settlements. The Guardian.
British Scientists Have Photographed Never Before Seen Sea Creatures
A surprising number of new sea creatures are being discovered. Even after centuries of scientific exploration, Earth is still full of surprises. Millions of species have been described so far, but thousands more continue to emerge each year, helped along by new tools like DNA sequencing that allow researchers to spot differences that once went unnoticed.
Read the full BBC report with stunning images. BBC.
Questionable Strategy to Reduce Ocean Warming by Dumping Chemical into the Sea
Boosting natural alkalinity using a chemical antacid should, in theory, encourage the ocean to absorb more carbon. Over a large surface area, and in combination with sharp emissions reductions, chemical treatment could prevent global temperatures exceeding 2C above preindustrial levels, while locally reducing ocean acidity, which is now higher than at any point in the past million years and poses a dire threat to marine life and fisheries. Full Details in the Guardian.
'
Suspended Ion Exchange
The Tampa City Council seems to be moving ahead with the design of a groundbreaking Suspended Ion Exchange (SIX®) technology at the David L. Tippin Water Treatment Facility (DLTWTF), set to become the largest of its kind in the world and the first full-scale SIX® system in the United States. Once completed, the facility will be capable of processing up to 140 million gallons of drinking water per day, a major leap forward in water treatment innovation. Smart Water Magazine.
Phoenix Wastewater Salvaging
With relentless heat literally melting away its water reserves, Phoenix has embarked on an ambitious program of wastewater salvaging. Arizona 15.
Tree Emits Holy Water
Residents of Pune, in India, gathered in large numbers to worship a tree believed to be emitting holy water. City officials eventually discovered that the water was actually coming from a broken water pipe that ran beneath the tree. India Blooms.
Fluoride in Birmingham
Residents of Birmingham, Alabama were informed that their drinking water would no longer contain fluoride. Then came a twist: It turned out that their water utility had, without telling the public, actually stopped adding it years ago. John Matson, a spokesperson for Central Alabama Water, which serves Birmingham and its surrounding suburbs, confirmed to NBC News that two of the utility’s four water filtration plants had temporarily stopped fluoridating in 2023, and a third had stopped in March 2024. The changes were made under prior leadership, he said, when the utility had a different name. NBC News.
Plastic Water Bottles
The authors reviewed 141 scientific articles to gather information about the scope of microplastic ingestion and associated health outcomes. Their findings were striking.
Across dozens of studies, the authors found that the average person might ingest between 39,000 and 52,000 plastic particles each year, “with bottled water consumers ingesting up to 90,000 more particles than tap water consumers.”
The conclusion Sajedi was concerning.
“Drinking water from plastic bottles is fine in an emergency, but it is not something that should be used in daily life. People need to understand that the issue is not acute toxicity — it is chronic toxicity,” meaning the dangers are primarily from extended regular use of beverages in plastic bottles that can add up over time. Yahoo.
The “Drought Paradox” and the Colorado River Basin
A new study reveals that during hot, dry periods, plants in the Upper Colorado River Basin are tapping into deeper groundwater reserves, diverting water from rivers and reservoirs. River flows in the region have already dropped by about 35% in recent years, with projections suggesting a potential 40% decline by mid-century, posing significant challenges for water supply in the American West. Intensifying drought conditions, driven by rising global temperatures, are exacerbating the issue, leading to water shortages that could impact food production, increase wildfire danger, strain local economies, and threaten public health. Yahoo News.
High School Student Invented a Filter That Eliminates 96 Percent of Microplastics from Drinking Water
Virginia teenager Mia Heller’s filtration system harnesses the power of ferrofluid, a magnetic oil that binds to microplastics in flowing water. Smithsonian
Water Story of the Month: Discovery Explains why Water Acts so Strangely
Scientists have finally found a hidden “critical point” in supercooled water that explains why it behaves so strangely. At this point, two different liquid forms of water merge, triggering powerful fluctuations that affect water even at normal temperatures. The breakthrough was made possible by ultra-fast X-ray lasers that captured water before it froze. This discovery could reshape our understanding of water’s role in nature—and possibly even life itself.
Researchers at Stockholm University have used advanced x-ray lasers to uncover a long-suspected feature of water: a critical point that appears when water is deeply supercooled. This occurs at about -63 °C and 1000 atmosphere. Even under everyday conditions, this hidden point influences how water behaves, helping explain many of its unusual properties. The results were published in the journal Science.
Water is everywhere and essential for life, yet it does not act like most other liquids. Properties such as density, heat capacity, viscosity, and compressibility respond to temperature and pressure in ways that are opposite to what scientists see in typical substances.
In most materials, cooling causes them to contract and become denser. Based on this pattern, water should reach its highest density when it freezes. Instead, ice floats, and liquid water is actually most dense at 4 degrees C. That is why colder water remains below warmer water in lakes and oceans.
When water is cooled below 4 degrees, it begins expanding again. If pure water is cooled below 0 degrees (where crystallization happens slowly), this expansion continues and even accelerates as the temperature drops further. Other properties, including compressibility and heat capacity, also behave in increasingly unusual ways as the temperature decreases.
Many excellent articles appeared on the subject in March.
|
|
Places to visit for additional information:
|
|
|
|
|
|