Pure Water Occasional, October 28 2017 |
In this late October Occasional you'll hear about cross connections, firefighting foam, sulfate in pig barn water, arsenic in baby food and domestic well water, toxic algae in clear lake water. Plus, drugs and tons of other chemicals in drinking water. Learn how utility company filters may be contaminating water. Read about artificial sweeteners as water contaminants and trees as nature's water filters. Find out how to "winterize" your big filter or softener and how to measure the flow of your well. And, as always, there is much, much more.
|
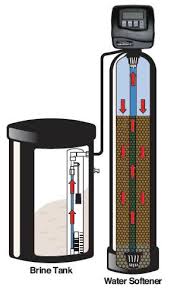
Winterizing Your Water Softener or Large Filter
|
If you keep your water softener or filter in the basement of an occupied home, you normally don’t need to worry about winterizing it. However, if the softener or filter is in an unheated garage, a vacation home that’s closed down for the winter, or a home that’s going to be left vacant during an extended winter vacation, you need to take precautions against freezing.
Insulating jacket for softener or filter tank.
If you live in a milder climate, where the weather doesn’t get extremely cold, insulating your pipes and tanks should be enough to protect your system during the winter. You can purchase pipe insulation wrap at any home improvement store. Heat tape or electric pipe heating cables for the water lines are also a good idea.
The tall tank needs protection from freezing; the shorter brine tank, probably not.
Some homeowners build an insulated box around the main media tank. You can also purchase plumbing insulation in sheets, or wrap an insulation blanket around the tank. There are even special jackets designed for water softener and filter tanks, or water heat insulation jackets can be modified to do the job.
Because of the salt saturation, your softener’s brine tank is only likely to freeze in very cold climates where temperatures stay above zero. After all, it has to get really cold for the ocean to freeze.
If you are using your water softener year round, the most important thing is to keep it warm enough to prevent freezing, which is why a space heater in your garage or another unheated space can help. Remember, you only need to keep the temperature above 32 degrees Fahrenheit.
Running water will also prevent freezing. If you are only going to be away for a few days, you could leave a faucet running at a slow trickle to keep things moving in the pipes. Although not a cure-all, this will at times prevent hard freezing and bursting from freezing pipes.
Draining and Disconnecting
If you do not plan on using your water softener during the winter, and the heat in your residence will be turned off during that time, there are specific steps you should follow to disconnect, drain, and store your system.
It is recommended that you drain the tanks. If there’s no water in the tanks, they can’t freeze.
Probably the easiest way to do this is to put the softener or filter into bypass mode to isolate it from your house plumbing, then disconnect it physically from the bypass valve so that the control valve can be screwed off the top of the tank.
With the riser exposed, you can use a 3/8″ tube inserted into the open riser to siphon the water out of the tank. You should be able to get almost all the water out of the tank. Although the water in the brine tank probably won’t freeze, it’s a good idea to dip out most of the water that isn’t actually covered by salt.
Unplug the control valve.
With all the water out of the tank, the filter or softener may actually be light enough to move to a warmer area. If it’s too heavy, leave it: it should be safe.
People with remote cabins who shut down seasonally might find it worthwhile to invest in a softener or filter built in a special “bottom drain” tank that makes the job of winterizing easy. These tanks can be drained completely without removal of the control valve or going to the trouble of siphoning.
|
Chemicals in Our Water
by Gene Franks
|
Since around 1980, when I first started paying attention to such things, the estimated number of different chemicals that find their way into our drinking water supplies has gone up. This is to be expected. What we should really be concerned about is our ability to monitor and regulate this onslaught of chemicals has not kept pace.
When the Toxic Substances Control Act (TSCA) became law in 1976 there were estimated to be about 62,000 chemicals in commerce. Under the provisions of the law, these chemicals were assumed to be safe unless the newly formed U.S. Environmental Protection Agency (EPA) found they posed an “unreasonable risk.” How many of these has the EPA studied since 1976? About 200. How many has it banned? Five. That’s five. With such oppressive over-regulation, it’s no wonder there are constant cries to reign in the EPA.
Now the estimated total has grown to 85,000 chemicals. Can anyone remember when the EPA last banned a chemical?
Every time water goes down the drain, whether to a sewer, septic system, storm drain or wherever, it carries contaminants with it, which usually end up in someone’s drinking water. Included are unmetabolized pharmaceuticals, chemicals, and particles from hand and face washing, bathing, laundry, the toilet—from virtually any and all human activity. The contaminants are in tiny concentrations, but from many thousands of sources and, as our use of pharmaceutical and personal care products (PPCPs) increases, our drinking water is becoming more contaminated.
The number of chemicals that surround us has grown beyond our comprehension. Mr. Cartwright says:
Globally, we now produce more than 85,000 different chemicals, many of which end up in our drinking water. Chemicals are used to manufacture 96 percent of consumer products; the average adult uses nine products per day containing 126 different chemicals. Fertilizers, pesticides, herbicides, and antibiotics are all also used in agriculture and animal husbandry operations. Whether from hand washing, bathing, showering, laundry, dishwashing, toilet use—no matter for what purpose we use water, it carries contaminants down the drain. If this water enters a municipal wastewater treatment system, it ultimately ends up in a body of water (lake, river, etc.), which often becomes a source of drinking water. If the wastewater is directed into a septic system, the treated water percolates into the earth, where it usually enters an aquifer or other water supply. Weather events generating runoff from lawn and agricultural surfaces also contribute to this contamination. It’s a fact of life: virtually every time water goes down the drain, it is carrying some contaminants that end up in someone’s drinking water.
Our drug habit, or consumption of “pharmaceuticals,” has gone far past what can be called epidemic proportions. A study in our local area of North Texas, a few years ago, demonstrated that estrogen spikes in our wastewater mirrored the schedule of two universities. Researchers found that when the population increased due to the schools being in session, the presence of estrogen also increased sharply because the chemicals were peed into the water. In relation, Mr. Cartwright writes:
Many of the pharmaceutical products we ingest are not completely metabolized, pass through the body, and contribute to [water] contamination. America is the largest pill-popping nation in the world, with 70 percent of us taking one prescription a day, 50 percent taking two, and 25 percent taking five or more per day. Additionally, because people are living longer, more pharmaceuticals are consumed and more end up in the water. Opioid addiction has become a crisis. In the US alone, in 2016, almost 4.5 billion medical prescriptions were issued. Other sources of contaminants include food, toothpaste, artificial sweeteners, caffeine, vitamins, as well as cosmetics, lotion, sunscreen, perfume, deodorant—the list goes on and on.
It would be foolish to assume that such vast chemical exposure poses no health risk to humans, although specific cause/effect relationships between chemicals and health are extremely hard to “prove.” Although Americans’ blood levels of glyphosate (RoundUp) keep going up, singling out the popular herbicide as a “cause” of any specific human ailment has eluded us.
Mr. Cartwright cites plenty of anecdotal evidence, however. A 2008 Canadian study showed that:
In 20 industrialized nations, the birthrate for boys has declined every year for the past 30 years. There has been a 200-percent increase in male sex organ abnormalities over the last 20 years. The average sperm count of North American college students has dropped by over 50 percent in the last 50 years. Up to 85 percent of the sperm in healthy males contains damaged DNA. Over the last 50 years, there has been a 300-percent increase in testicular cancer. For many years, there have been reports of feminization in fish and amphibians, as well as documented genitalia deformities in such diverse animal populations as bears, panthers, sea lions, whales, birds, alligators, and others. Between 1999 and 2003, in a population of Chippewa aboriginal peoples in southwestern Ontario, Canada, the birth ratio of boys to girls declined from roughly 50/50 to 33/67.
On chemicals known as endocrine disrupters (EDCs) alone there has been extensive research which clearly reveals the association of exposure to EDCS and human disease. Mr. Cartwright quotes the journal Endocrine Reviews:
"Whether low doses of EDCs influence certain human disorders is no longer conjecture, because epidemiological studies show that environmental exposures to EDCs are associated with human diseases and disabilities." A follow-up review in 2015 contains the statement: "It simply is not reasonable to assume a chemical is safe until proven otherwise. Clearly, not all chemicals are EDCs, but substantial information needs to be provided before inclusion of a new compound in a food-storage product, a water bottle, health and beauty products or a household product."
A significant hindrance to establishing clear cause/effect relationships with chemicals is that EDCs can have even transgenerational effects. It is not easy to trace a person’s ailments back to chemical exposure of his or her grandfather, for example.
Transgenerational effects of EDCs mean that even if a chemical is removed from use, its imprints on the exposed individual’s DNA may persist for generations and possibly forever.
Other common chemicals we are regularly exposed to via drinking water include disinfectants and additives like fluoride.
Chlorine, the common water disinfectant used in municipal drinking water treatment plants can chemically react with some PPCPs and produce DBPs, a class of trihalomethanes (THMs) containing chemicals known to cause cancer. In recognition of this, the U.S. EPA has established a maximum limit for THM compounds, listed in the Safe Drinking Water Act. Many municipalities are adding ammonia to chlorine to produce chloramines, which do not generate dangerous DBPs. The formation of these compounds is an example of the complex chemistry associated with PPCP contamination.
Then there are the “emerging contaminants” known as PFAS.
In addition, fluorine-based chemical contamination of aquifers has become a major issue in many areas. Under the general acronym, PFAS (poly- and perfluoroalkyl substances), they are major components of firefighting foam, Teflon® and Scotchgard® products, coatings on carpeting, clothing, fast-food wrappers, and many other consumer products. PFAS exposure has been linked to cancer, obesity, immune system suppression, and endocrine system disruption.
Today, multiple agencies, including the EPA, the CDC, and the U.S. Deptartment of Health and Human Services, face the seemingly hopeless task of keeping up with and reporting on the effects of chemicals on the U.S. population. The EPA, for example, publishes a report every two years. The most recent (2017) report included data on 308 chemicals. Let’s see–308 of how many? 85,000. Obviously, chemicals are being put into the environment a lot faster than regulators can regulate them.
Nevertheless, there is, as Mr. Cartwright notes, “a continuous stream of news releases on credible scientific studies that address links between common household chemicals and various health effects.” So many that we can’t keep up.
Here are a few:
• In a 2014 study at Columbia University, two chemicals found in such products as lipstick, hairspray, nail polish, dryer sheets, and vinyl fabrics (phthalates: suspected EDCs) lowered the IQ of children born to mothers exposed to them.
• A recent Virginia Tech study has found a connection between quaternary ammonium compounds (quats) found in cleaners, laundry detergent, fabric softener, shampoo, conditioner, and eye drops, and birth defects in laboratory rodents.
• Again, common household products are implicated in a Washington University in St. Louis study that linked them with ovarian function, resulting in women experiencing menopause two to four years earlier than normal.
It is very important to underscore the fact that, so far, there is no proven link between these trace contaminants and human health. Although many scientific studies are underway, there is lack of conclusive proof that PPCPs are harmful. On the other hand, with so many different chemicals in our drinking water (in this writer’s opinion), it is only a matter of time before a health risk is identified.
Mr. Cartwright’s research points to many unanswered questions involving the relationship of waterborne chemicals with cancer, autism, ADHD, Parkinson’s disease, diabetes, allergies, and more. He suggests the most vulnerable populations are babies, the elderly, pregnant women, and adults with compromised immune systems. It is unclear if the most dangerous chemicals are those that bioaccumulate in the body or those that break down in the body. And he asks probably the most persistent and most difficult questions: Are there combinations of chemicals that present greater risks than individual chemicals? Do these chemicals react with each other to produce other dangerous compounds?
|
Measuring Flow Rate of Wells |
The flow rate capability of your well should be measured accurately because many backwashing water filters require an adequate flow rate to keep the media clean. Timing how long it takes to fill up a measured bucket is an inaccurate method of attaining flow rates unless you have a “constant pressure” well that delivers water at a more or less fixed rate. For conventional pressure tank wells, the single-shot bucket method is not accurate.
The proper well water flow rate is determined by counting the gallons drawn down and the time between cut in and cut off cycle of the well pump. To do this, you’ll need some kind of timing device, like a stop watch, plus a container of known size to catch the water.
1. Allow the well pump to build to full pressure. Shut off the main water valve to the building to assure that no water is being used.
2. Then, open a spigot below the pressure tank, capture the water, and measure the number of gallons drawn down from the pressure tank until the well pump turns on. You can measure in a small bucket, because it’s OK to turn the water off while the bucket is being emptied.
3. When the pump turns on, immediately close the spigot and time the period it takes for the well pump to recover. That is, see how much time lapses between when the pump turns on and when it turns off.
When you have this information, the formula for determining the flow rate is gallons drawn down that were measured above, divided by the seconds required for recovery, then multiplied by 60.
(Gallons / Seconds) x 60 = Gallons per Minute (gpm) flow.
For example, if 16 gallons are drawn down and it takes 90 seconds to build pressure back up, then: 16 divided by 90 = .177 gallons per second.
Consequently, .177 x 60 = 10.6 gallons per minute flow rate.
What you are calculating is the sustained flow rate of the well. In other words, the gpm rate that the well can put out over the time necessary to backwash a filter. This can differ considerably from the “first bucket out” rate taken when the pressure tank is full.
Backwashing filters need sustained flow for several minutes to complete their cycle, and a filter should not be installed on a well that will not supply enough gpm flow to backwash it.
|
How Trees Help Keep Water Clean
by Lin Taylor
|
LONDON (Thomson Reuters Foundation) – More trees at water sources improve sanitation and lead to fewer children dying from diarrhea in poor countries, a global study said on Monday.
The study examined the health of 300,000 children and the quality of watersheds across 35 countries including Bangladesh, Nigeria, and Colombia, and found that having more trees upstream led to healthier children.
“This shows, very clearly, how healthy ecosystems can directly support human health and welfare,” said co-author of the study, Brendan Fisher, a researcher at the University of Vermont in the United States.
“This suggests that protecting watersheds, in the right circumstances, can double as a public health investment,” he said in a statement.
The study, which analyzed U.S. foreign aid data spanning three decades, said increasing the number of trees by a third near the source of watersheds in rural areas could improve water sanitation as effectively as installing indoor plumbing or toilets.
“These findings clearly show that forests and other natural systems can complement traditional water sanitation systems, and help compensate for a lack of infrastructure,” said Diego Herrera, lead author of the study which was published in the journal Nature Communications.
Diarrheal disease, which is preventable, kills more than half a million children under the age of five each year, and is one of the leading causes of death in children along with malaria and pneumonia, the World Health Organization (WHO) says.
Lack of sanitation and clean water result in nearly 1.7 billion cases of childhood diarrheal disease every year, the WHO says.
|
Additional Facts about Trees and Water
|
Most Spruce trees are, by weight, about 20% water.
|
You've probably seen the charts that show how much water is contained in the makeup of everything from an orange to the human heart. Massive, solid trees are no different. Their mass is comprised of a significant amount of water. And like people, not all trees are equal in water content.
The Catalpa tree in my back yard, according to one source, is, by weight, 48.25% water. Trees listed range from 7% to 54% water.
|
Artifical Sweeteners are Clogging Ontario's Grand River
by Ivan Semeniuk, Science Reporter
|
Ontario’s Grand River is so chock full of artificial sweeteners that scientists say the chemicals can be used to track the movement of treated waste in the region’s municipal water supplies.
Artificial sweeteners are used as sugar substitutes in diet drinks and foods. They impart no calories because they are not readily broken down in the human digestive system, so they tend to exit the body intact. But that persistence also means the sweeteners linger long after they are flushed away. They survive processing in waste-water treatment plants, find their way into the environment, and reappear in drinking water.
As part of a long-term study, scientists with Environment Canada and the University of Waterloo repeatedly sampled 23 sites along the Grand River system as well as household taps. Four artificial sweeteners – acesulfame, saccharin, cyclamate and sucralose – were detected, in some cases at higher concentrations than reported anywhere else in the world.
At one site, the researcher calculated the equivalent of 90,000 to 190,000 cans of diet soda were being consumed each day to account for the quantity of acesulfame they measured. “If you think about all those cans of pop floating down the river, it’s quite an image,” said Sherry Schiff, a biogeochemist at the University of Waterloo and a co-author of the study, published Wednesday in the open access journal PLOS ONE. Nearly one million people live in the region, which includes the communities of Kitchener-Waterloo, Guelph, Cambridge and Brantford. About half of those people rely on the Grand for their drinking water.
While the sweeteners are approved for human consumption and are present in far greater concentrations in diet products, their appearance in drinking water may come as an unnerving surprise to some. The main aim of the study was to see how well the chemicals can be used to keep tabs on where wastewater ends up. Because of its stability, acesulfame in particular “seems to be perhaps the most ideal tracer of wastewater found so far,” said John Spoelstra, a research scientist at the National Water Research Institute in Burlington, Ontario, and lead author on the study.
Armed with such a tracer, he added, scientists can better spot where human waste is coming into a watershed – not only from municipal water treatment plants, but from septic systems that may be leaking into groundwater. The changing concentrations of the sweeteners can also be compared with that of pharmaceuticals or other chemicals that may pose a health risk but whose pathway and evolution in the water system is not well understood. “We need a tracer to be able to distinguish dilution effects from actual removal and degradation effects,” Dr. Spoelstra said. He added that while other researchers have looked at artificial sweeteners in drinking water systems, this is the first time a study has documented their movement on the scale of an entire watershed.
“We’re looking at how sweeteners are accumulating in that water as you move downstream and more and more wastewater is coming into the system,” Dr. Spoelstra said.
The authors stressed that more research is needed to understand the long-term impact of the sweeteners on the environment. Acesulfame, for example, can break down under exposure to ultraviolet light from the sun, producing compounds that are more toxic than the sweetener itself.
The Grand River is the largest Ontario River that drains into Lake Erie.
|
Here are just a few of the many articles about water issues that are available every day from the Pure Water Gazette website. |
Pigs resistant to sulfate in water
Studies reported in the Journal of Swine Health and Production demonstrate the capacity of weanling pigs to handle large amounts of sulfates in their drinking water. Pig performance was not adversely affected by poor water quality. Issues with poor growth, feed intake, or diarrhea should not be attributed to water quality problems without first exploring other influences, such as environmental, social, and nutritional stresses imposed at weaning. Certainly, observing pigs on high sulfate water with diarrhea should not automatically lead to the conculsion that they are performing poorly. Sulfate in the water causes an osmotic diarrhea, which is very different from a secretory diarrhea that might result from pathogens. Based on a large number of studies of various sizes and conducted under a variety of conditions, it appears that unless water is of extremely poor quality, meaning it contains more sulfate than observed in this study, the likelihood of it resulting in poor animal performance is quite low. The moral: don't bother to get your pig a reverse osmosis unit if your water is high in sulfate.
California denied federal funding for water project
The Trump administration pulled support from Governor Jerry Brown's ambitious plan to build California's biggest water project in decades, casting the current form of the $16 billion proposal to build two giant tunnels as another unwanted legacy from the Obama era.
Baby food tests positive for arsenic
An alarming study found many baby food products test positive for arsenic, including 80% of infant formulas. Mainstream brands including Gerber, Enfamil, Plum Organics, and Sprout were among the worst offenders.
Large Gulf oil leases may become available for purchase
The Trump administration is proposing that nearly 77 million acres in the Gulf of Mexico be made available for companies wanting to purchase federal oil and gas leases; the largest offering ever made in the United States.
Arsenic present in U.S. wells
It is estimated that high levels of arsenic in domestic wells could affect as many as 2 million people in the United States.
Senate bill will provide funding for research on toxic algae blooms
U.S. Senator Charles Schumer, alarmed by the reappearance of toxic algae in Skaneateles Lake, urged House members to quickly pass a bill that would provide $110 million in emergency funding to fight a growing problem in upstate New York. The Senate passed a bipartisan bill September 26th that would authorize $22 million a year for five years, paying for research and response efforts to prevent blooms of toxic blue-gree algae on lakes, ponds, and sources of drinking water across the nation.
Beware of clear lakes
Scientists who observed lakes in intensively agricultural areas have concluded that clear water does not necessarily mean the lake is free of excessive nutrients. In fact, they found some lakes are so excessively fertilized most of the algae and cyanobacteria containing the green pigment chlorophyll were killed. Water quality in these lakes was found to be worse than in lakes whose water looked like pea soup.
Are utilities' filters contaminating tap water?
A North Carolina State University research team is testing water in New Hanover and Pender counties after tests indicated potentially dangerous compounds could be leaching from utility filtration devices even after discharges cease upstream. The study is based on the fact that levels of certain contaminants (GenX and six other perfluorinated compounds) in the lower Cape Fear River have persisted in treated water long after levels in the raw water have decreased.
Follow daily water headlines and links to full articles from Environment Health News at the Pure Water Gazette.
|
Places to visit on our websites |
Thanks for reading and be sure to check out the next Occasional!
|
|
|
|  | |