Pure Water Occasional, November 13, 2018 |
In this early post-election Occasional, you'll learn about how squeaky clean skin isn't necessarily a good thing, why nutria and black wattle are enemies of water, how to change the media in a water filter, why Mexico City is shutting off its water, how the EPA has affected blood lead levels in children, where softeners rank on the salt pollution index, and how to run a softener on potassium chloride. Then, there are PFAS (not PFASs or PFCs), acronyms, Austin's boil water alert, environmental regulation rollback, filter carbon, Dosatron, and, always, much much more.
|
There are indications that women are more concerned about water issues than men, based on analysis of polling at Texas A&M University. Women reported greater average concern on every water issue they were asked about. The biggest differences, compared to men, were in areas of water loss, climate change, conservation and affordability. One conclusion drawn from the study: “The path to building support for water systems in American communities probably starts with women.”
We recently reported on goldfish and zebra mussls as invasive species that damage water systems. Here's another: In California, officials announced the capture of the 300th nutria (aka coypu) in the state and suspect several breeding populations may exist. The large rodent, native to South America, can damage levees and irrigation canals and alter habitat.
Study finds water softeners contribute to salty water bodies
A new study from the University of Minnesota found that home water softeners are sending a significant amount of salt into the environment. Researchers from the University’s Water Resource Center created a “chloride budget” to estimate how much salt enters the environment each year from different sources. While road salt was found to be the largest contributor, home water softeners were fourth on the list.
The study found that overall in the state of Minnesota road salt contributes more than 400,000 metric tons of chloride annually to the environment, while home water softeners contribute approximately 140,000 metric tons of salt per year.
The study found while most Minnesota water treatment plants are not equipped to remove chloride from water, home owners can take steps to mitigate the environmental impact of their water softeners. The report suggests water softeners should be serviced regularly and be set up based on the hardness level of the water and not an automatic timer.
Hope for the survival of the Clean Water Act
Former U.S. Environmental Protection Agency (EPA) administrator Gina McCarthy thinks there’s little chance the Trump administration efforts to roll back environmental regulations will succeed, according to an October 9, 2018 article in Quartz.
In spite of the Trump administration’s announcements about rolling back various environmental regulations, McCarthy said that they cannot be revoked until they are replaced with new rules. Regulations put in place during the Obama administration, such as the Clean Power Plan, were “solidly done,” she said. “They were based on science and the law and I think it’s going to take a lot to undo them.” The courts have already rejected several Trump administration environmental proposals and the trend is likely to continue, she said.
Other news items:
Perfluorinated alkylate substances (PFAS) have existed for decades and are used in hundreds of everyday products, but only recently have scientists begun to understand the negative health effects associated with this class of industrial chemicals. Research recently published by Harvard University's Philippe Grandjean indicated that some companies were aware of the risks these chemicals posed in the late 1970s but did not share the findings with the EPA until decades later.
Regulation matters: The average amount of lead found in the blood of children living in the United States was 16 micrograms per deciliter in 1976-1980. It is now down to less than 0.84 micrograms per deciliter mostly due to EPA regulation of leaded gasoline.
Baltimore voters made history in the November election by passing a city charter amendment banning the privatization of the city’s water and sewer systems. Baltimore is now the first major city in the country to amend its charter to prohibit the sale and lease of the city’s water and sewer system.
The Austin, TX municipal water supply was under a boil water alert for several days in October due to excessive particulate caused by heavy rains and flooding. City officials credited the rapid return to normal service to excellent cooperation from citizens who voluntarily cut back water use to give the treatment plant time to catch up.
After years of insisting its water is safe, Newark, NJ has begun "an urgent giveaway of some 40,000 water filters across the city." The problem is the same as with Flint: lead service lines. The source water and the treated water test fine, but the ancient infrastructure is putting lead into the water delivered to homes. Story.
Megacorte, the Great Water Cut-off
Mexico City scheduled a complete cutoff of city water in early November. In anticipation, residents filled every container in sight and many businesses planned to close for the entire 72-hour period. Workers are making much-needed repairs to Mexico City's so-called Cutzamala system. It is a 200-mile-long complex water delivery system composed of several huge lines, reservoirs and pumping stations that send water up and over the mountains surrounding Mexico City.
Water service was shut down in the western Washington town of North Bend after it was discovered someone had broken into the town's water tower and small white beads were found on the tower lid and surrounding area. The pellets were tentatively identified as "biodegradable airsoft BBs" (not a major threat to water quality), but the tower was drained and cleaned just to be safe.
When plants move outside their native region they can become great water hogs. A Nature study reveals that invasive plant species in South Africa "take a shocking toll on the water supply."
This year, Cape Town, South Africa almost became the first major city in the world to run out of water. (It was saved at the last minute by stringent water restrictions). In May, researchers argued that alien plants, which often use more water than do indigenous ones, consumed more than 100 million liters of water a day — about a fifth of the city’s daily usage. They warned water losses due to invasive species could triple by 2050 because trees including black wattle and cluster pines are spreading. The latest report estimates that invasive trees and shrubs, if left unchecked, could threaten up to a third of the water supply to cities such as Cape Town, and consume up to 5% of the country’s mean annual rainfall runoff.
A southern Arizona woman accused of fraudulently altering water sample test results for water providers that were clients of her company has pleaded guilty to attempted fraud. The 67-year-old woman agreed to a plea agreement which calls for her to serve eight months in jail and pay a $10,000 fine. The charge accuses her of changing water testing paperwork submitted to state regulators to replace positive results for bacterial contamination with negative results.
An investigation by the FDA and CDC concluded that E. coli infestation of romaine lettuce which killed five people and caused illness in many others could be traced to tainted water from an irrigation canal. The incident resulted in related illness reported in 36 states and was the worst E. coli outbreak since 2006.
|
Dosatron NSF Certified, Water Powered
Chemical Injector
|
Dosatron has the only water powered chemical injector that is NSF/ANSI 61 & 372 Certified. The NSF-certified 14 gallon per minute Dosatron D14 unit is ideal for injection of water treatment chemicals like chlorine and soda ash in residential applications.
Water-powered pumps offer several advantages. They are very easy to install, require no electricity, and feed the injected chemical proportionally, depending on the rate of flow through the pipe. This means they can be installed at any place in the water line without flow switches or the expensive metering equipment required with electric pumps installed after the well’s pressure tank.
The fully adjustable D14 Dosatron injects at a flow rate of 1:500 to 1:50. It is a compact pump that installs directly into the water line. As water runs to the point of use, the pump injects the water treatment chemical into the line. It can be used to disinfect non-potable water or to pre-treat for iron, manganese, and hydrogen sulfide filters. The pump is so light that it can be supported by the pipe itself, or it can be wall mounted (bracket is included) and installed with hose connections.
Dosatron offers an easy way to add chlorine or soda ash treatment. It is durable and easy to service, and parts are readily available.
Water driven pumps are ideal for off-grid applications or any remote location where electricity is not available. Dosatron is ideal for feeding chlorine (regular household bleach) in front of iron, manganese, or hydrogen sulfide filters, or for making non-potable water safe to drink. The D14 works in any application where the water being treated runs 14 gallons per minute or less.
The D14WL2NAF is the NSF-certified, drinking water grade of Dosatron units. It should not be confused with Dosatron models intended for agricultural use that are sold on many websites.
|
50 years later, Sioux Falls manages contaminated water from toxic firefighting foam
|
Gazette Introductory Note: We’re reprinting this piece to illustrate the widespread problem with PFAS and to show how one water supplier has chosen to deal with it. Municipal treatment of this growing chemical threat to drinking water is difficult and expensive, to say the least.
Sioux Falls, SD officials are grappling with well shutdowns as the extent of the city’s water contamination from decades of firefighting foam use remains unclear.
Sioux Falls currently has 19 municipal wells sitting dormant in the aftermath of innumerable gallons of toxic firefighting foam that contaminated the grounds of the city airport nearly 50 years ago, the Rapid City Journal reported. Chemicals linked to cancer and other health issues were found to have contaminated 15 municipal wells, including 10 that have concentrations above what the Environmental Protection Agency deems safe.
About 28 percent of the city’s water production from the Big Sioux aquifer is shut down.
The South Dakota Air National Guard and the Sioux Falls Fire Department both used the toxic firefighting foam for many years near the airport, which led to the contamination of the city’s drinking water. But the scope of the issue is still unknown.
“We really haven’t determined the extent of release yet,” said Capt. Jessica Bak, a public affairs officer with the Air Guard at the Sioux Falls Regional Airport.
In 2013, the city’s water purification plant found chemicals from firefighting foam, known as per and polyfluoroalkyl substances (PFAS), at levels below the EPA’s health advisory level. The level of exposure beneath the EPA’s threshold means there aren’t expected adverse health risks.
The city responded to the findings by testing all municipal wells to identify the source and shutting down every well where the chemicals were found.
City engineer Tim Stefanich, who oversees the water system, acknowledged that “there was a little bit of time between” finding the contamination, determining its source and deciding to shut off wells. But he said that there was minimal fear of an immediate health risk with the low levels of exposure.
The city tested for PFAS again in 2014 as part of an EPA-mandated water sampling program, but didn’t detect any of the chemicals. The city tested again in 2016, when some low levels were found.
The city shut off more wells, leading to the 19 wells offline today. Water leaving the city’s purification plant is now sampled monthly, and no water samples have contained the chemicals since 2016.
Stefanich and Trent Lubbers, the city’s utilities operation administrator, believe the contaminated water situation is under control.
The city has been purchasing water from the Lewis and Clark Regional Water system, a nonprofit, wholesale provider of treated water. But, Sioux Falls will likely need a more sustainable option.
“They have the short term kind of covered,” said Mark Meyer, drinking water program administrator for the state’s Department of Environment & Natural Resources. “But as we march into the future, having 28 percent of their well capacity offline, the future is going to come sooner than later.”
Reprinted from Argus Leader
|
Filter Carbon: What It Does and What It Doesn't
by Gene Franks
|
If you’ve wondered what makes water filters effective, this article will give some answers. Water filters aren’t magic. Except for rare specialty items, what we call a water filter works because of a single ingredient–activated carbon. The present article is a slightly truncated and revised version of one that’s been on Pure Water Products’ main commercial website for a number of years.— Hardly Waite, Editor.
The largest single section in the “EPA Regulated Water Contaminants” list is the section on Organics (including VOCs, or “Volatile Organics”). In this category the EPA lists numerous very nasty organic chemical contaminants—many with familiar names like benzene, 1,1 dichlorethylene, carbon tetrachloride, dioxin, styrene, toluene, chloroform, and vinyl chloride. To give an idea of the extensiveness of this list, a single item called “Total Trihalomethanes” consists of hundreds of chemicals, some still undiscovered or unstudied, that are formed as by-products of the chlorination process. The EPA’s maximum allowable level for trihalomethanes, many of which are suspected or known cancer causers and are present in virtually all chlorinated tap water, is less than 1/10 of one part per million.
For the Organics category, the primary treatment in all cases, and the only recommended treatment in most cases, is activated carbon.
The EPA’s Pesticides category lists many familiar poisons such as Aldicarb, Chlordane, Heptachlor, and Lindane. In all cases, activated carbon is the only recommended treatment.
Of the Herbicides listed (2,4-D, Atrazine, etc.), activated carbon is the only treatment recommended.
In short, for Organics, Pesticides, and Herbicides, which together make up 90% of EPA’s regulated substances, the standard treatment, and in many cases the only treatment recommended, is activated carbon.
What carbon filtration does not do can be seen in the remaining three categories of the EPA contaminant list.
Microbiological contaminants. Carbon is listed as a treatment only under one category–turbidity. It is not recommended for coliform removal or for cysts, though in reality some of the very tight solid carbon block filters now on the market remove bacteria (though manufacturers seldom make this claim), and many carbon block filters are now certified for removal of cysts (giardia and cryptosporidium).
Inorganic contaminants. Carbon appears in the EPA list only as a preferred treatment for mercury. Carbon filters are also frequently engineered to remove lead (by the addition of ion exchange resins) and some carbon filter makers claim asbestos reduction, but by and large carbon is not a good treatment for most inorganic water contaminants.
The same is true in the final category, Radionuclides, where carbon is ineffective and reverse osmosis (RO) and ion exchange are definitely the treatments of choice.
Chloramines and Chlorine
Chlorine was not considered in the discussion above because the EPA does not consider it a water contaminant. Nevertheless, chlorine removal is a top priority of water filter purchasers. Chlorine removal is what carbon is best at, and nothing else equals carbon’s ability to remove chlorine. Carbon, especially the specially processed carbon known as “catalytic carbon,” is also the best treatment by far for chloramines, the increasingly (un)popular chlorine substitute.
Fluoride, another EPA-unlisted additive that people frequently want removed from tap water, is not readily removed by carbon filtration. (Although carbon can remove fluoride under the right circumstances, its performance is sporadic and unpredictable, so it’s best not to count on it.) I should note, too, that a unique carbon called “bone char,” which is made from animal bones, is widely used as a fluoride remover in some parts of the world, though rarely in the United States.
TDS, “total dissolved solids,” the count of the total mineral content of water, is not affected by carbon filtration. The EPA suggests a non-enforceable upper limit of 500 parts per million (ppm) total dissolved solids. TDS can be reduced only by reverse osmosis or a distiller or by a very expensive process called deionization.
Sodium, Fluoride, and Nitrates
Tap water ingredients people most frequently want removed and are not readily removed by carbon filtration are fluoride, nitrates, and sodium. (Sodium is measured in water as part of the Total Dissolved Solids, discussed above.) Reverse osmosis and distillation remove all three. Either of these methods, combined with a high quality carbon filter, provides complete treatment. All three contaminants can also be removed by selective, non-carbon filters designed for the purpose. For example, you can obtain a double filter with one fluoride and one carbon cartridge, if fluoride removal is desired. The fluoride cartridge does not contain carbon but a specialty medium called Activated Alumina.
When distiller sellers or zealous home marketers show you a chart indicating that reverse osmosis (RO) units do not remove chlorine or certain chemicals, keep in mind that RO units contain one or more carbon filters. In fact, “thin film” RO units, the most common type, must remove chlorine from the water as the very first operation else the unit’s membrane will be destroyed. Such statements are simply advertising cheap shots that are technically true but in reality totally false and intentionally misleading. It’s surprising and disappointing that some large companies actually do this in their promotional literature.
A reverse osmosis unit that has at least two high quality carbon filters is the best and most complete drinking water treatment for the home. When people say they want a water treatment system that “removes everything,” reverse osmosis is as close as you can get.
|
WQRF issues RFP for study on contaminants between MCL and MCLG
by Gene Franks
|
When I saw the headline above I knew I was going to have to learn some new acronyms. Just last week we put up an article about the difficulty writers and readers and researchers are having with the many new abbreviated forms used for “emerging contaminants” that start with “P.” The world is being overrun by acronyms, and the water treatment industry creates way more than its share.
WQRF, I learned, stands for the Water Quality Research Foundation, which was formerly called the Water Quality Research Council (WQRC), which was formed in 1949 to serve on behalf of the Water Quality Association (WQA) as a universally recognized, independent research organization.
Then, I learned from the Wikipedia that RFP stands for “request for proposal.” The RFP is a “document that solicits proposal, often made through a bidding process, by an agency or company interested in procurement of a commodity, service, or valuable asset, to potential suppliers to submit business proposals.”
I already knew what MCL and MCLG mean, but to be sure I understood them in the context of the WQRF’s RFP, I looked them up:
MCL stands for Maximum Contaminant Level: the highest level of a contaminant that is allowed in drinking water. MCLs are set as close to the maximum contaminant level goals (MCLG) as feasible using the best available TT (Treatment Technology).
MCLG stands for Maximum Contaminant Level Goal: the level of a contaminant in drinking water below which there is no known or expected risk to health. MCLGs allow for a margin of safety.
Here’s how the project goal itself is stated in the article:
This study is the first attempt at collecting and analyzing national occurrence data between the MCL and MCLG, utilizing data that is available from state and federal databases including, but not limited to: EPA, CDC, USGS, FRDS, NCOD, and SDWIS. Of the contaminants governed by the National Primary Drinking Water Regulations, only those that have a MCLG value lower than its MCL value (including MCLG values of “zero”) will be included in this research.
FRDS, NCOD, and SDWIS, which I didn’t understand, I decided to leave it alone. You don’t have to know everything.
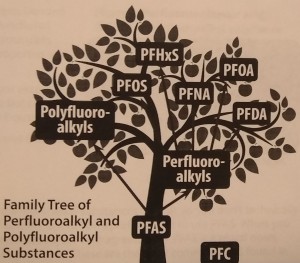
In regard to the “P” word contaminants that we went to so much trouble trying to classify, this very week, the Agency for Toxic Substances and Disease Registry (ATSDR), apparently a division of the CDC (Centers for Disease Control), seeing the urgent need to get everyone on the same page so these chemicals can be talked about, issued a very helpful document called The Family Tree of Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS) for Environmental Health Professionals. I hope you’ll read it. It keeps things simple by showing only the main PFAS family and leaving off the subfamilies. It also drops one confusing acronym, PFC, from the tree, pointing out that PFC stood for perfluorinated chemicals and also for perfluorocarbons. They do not mention that it also stands for Private First Class, which probably confused lots of people. PFC shows on the picture above as a fallen apple.
One very edifying part of the Family Tree, though, is the clarification of the singular/plural issue. I learned I’ve been making some pretty dumb statements (as have most of the people who write about PFAS). According to the ATSDR, PFAS is plural, so you shouldn’t add an “s” to it and write PFASs, as many, including me, have done. Putting an “s” on PFAS is like saying, “My uncle has three childrens and they all wear red hatss.”
We live and learn.
Pure Water Annie always crosses the t’s and dots the i’s and never puts s’s on plurals.
|
Water Softener FAQ: Regenerating a Softener with Potassium Chloride
by Pure Water Annie
|
Gazette Technical Writer Pure Water Annie Answer’s the Persistent Questions about Water Treatment
|
|
Why do some people use potassium chloride rather than salt to regenerate their water softener?
The main reason is to avoid using sodium – for health or environmental reasons. In fact, some cities require its use in an effort to limit sodium levels in wastewater.
How does the cost compare with standard softener salt?
Potassium chloride (KCl) is more expensive; considerably more expensive in some areas. Also, salt is more readily available. Since potassium is used in farm fertilizer, it’s in much shorter supply and it may eventually get very expensive.
I’ve heard you have to use more potassium chloride than standard softener salt to regenerate the softener. Is this true?
In theory, it would seem to be true, but research has shown this isn’t a big factor. In one study, when using 15 lbs. of salt per cubic foot of resin for regeneration, standard salt was only about 2% more efficient than potassium chloride.
Are there problems with potassium chloride?
Other than the cost and in some areas the lack of availability, one issue is the tendency under certain conditions (mainly temperature swings when softeners are installed in unheated areas) for a condition called recrystallization to occur. This is similar to the condition called “salt bridge,” with regular salt, but when temperatures fluctuate potassium chloride can precipitate and drop to the bottom of the brine tank forming a solid mass beneath the salt grid platform. There are strategies to alleviate this which include insulating the brine tank or elevating the tank on a wooden platform.
I have a reverse osmosis drinking water system. They told me it takes the sodium out of my softened water. Is this true?
Yes. It will also remove potassium, so if you have an RO unit for drinking water, your choice of a regenerant has no health implications.
And if I don’t have an RO unit?
The amount of sodium you ingest in softened water is pretty insignificant when compared with your total sodium intake, so drinking softened water normally isn’t important. For people trying to eliminate all sources of sodium, of course, it may be a different story.
If I want to try potassium chloride in my softener, do I need to make modifications?
No, salt and potassium chloride both work well in standard water softeners.
|
Changing the Filter Media in Home Water Filter Tanks: The Hard Part Is Getting the Old Media Out
|
|
Home tank-style water filters, either backwashing filters or simple “in/out” units that do not backwash, are essentially trouble-free performers, but eventually, usually after a few years of service, the filter medium inside the tank has to be removed and replaced. Removal is usually the hard part of the job.
Professional water treatment dealers often have special tools and tricks to make media change easy. They also may have a couple of muscular young guys who are used to dealing with heavy tanks.
Here are a few tricks for homeowners, who usually have none of the above.
If the filter is small, and if it is installed in an easily accessible location, the job can be pretty easy. If the filter is large–like a 13″ X 54″ tank, for example–and/or if it is installed in a hard-to-reach spot, the job won’t be easy.
After you have turned off your water supply to the filter and disconnected it from your plumbing, you can open the filter by simply screwing the control head off of the tank. It’s like a lid on a bottle. No tool is needed, but if it’s on tight it may be a two person job, with one holding the tank and the other screwing off the control head. Tip: If your filter has a Fleck 2510 control valve, don’t remove the clamp just above the lip of the tank. Screw the entire head, clamp and all, out of the tank.
With a small tank that’s easily accessible, the best strategy is to muscle the tank outdoors or to a floor drain, invert it and dump out the water and media inside. If you’re outdoors, it’s very easy to tilt the tank to a 45 degree angle and insert a garden hose, with water running, into the tank to rinse out the media.
However, if the tank is large and heavy, and especially if it is installed in a cramped space where it would be hard to remove, a suggested option is the use of a small wet vacuum, often called a “shop vac.” Using a shop vac you can suck the water and the spent media from the tank without moving the tank.
Depending on the size of your shop vac and the size of your filter tank, you’ll have to empty the shop vac a few times to accomplish the task. It can be a messy job, but it’s much easier than moving a heavy filter tank.
Before you start the job, be sure that the vacuum cleaner’s wand will fit into the mouth of the tank. Standard tanks have a 2.5″ opening at the top. You may be able to pull the center tube (riser) out of the tank to make the job easier. However, if you have a “Vortech” style tank with a fixed riser, you’ll have to work around it. With a fixed-riser tank, you’ll need a vacuum wand that’s no larger than 1.25″ in diameter. As the picture shows, you can nudge the riser to the side to get the vacuum wand into the tank.
A 1.25″ vacuum wand will fit even into tanks where the riser tube cannot be removed.
When the tank is empty, you may want to rinse it by using a garden hose if you have it outdoors or by adding water and then vacuuming it out, if you’re tank is being serviced in place. It is normally not necessary to sanitize the tank before adding media, but if you want to, add a couple spoonfuls of household bleach, refill with water, and let it stand for a few minutes before removing the water.
When the tank is ready to reload, if you have a removable riser reinsert it into the tank and be sure it goes all the way to the bottom and stays there while you pour in the gravel underbed and the media. (No gravel is needed in fixed-riser Vortech tanks, like the one in the picture.)
A useful tool for filling the tank is a media funnel; if you don’t have one, you’ll have to improvise. Before you pour in the media, be sure to close off the top of the riser with tape or a small plastic bag to prevent media from going into the tube. Pour in the gravel (if needed) first, then the media. In most cases you’ll end up with a tank that’s about 2/3 full.
When the media is loaded, lube the o-ring at the bottom of the control valve and the o-ring inside the center hole of the valve where the riser tube will slide with silicone grease. Screw on the head securely, but don’t over-tighten. Reconnect your plumbing and you’re ready to go.
|
Does Hard Water Make Your Skin Squeaky Clean?
|
Common complaints about soft water, either naturally soft or water softened by water treatment, are that soft water leaves the body with a slick, slimy feel, that soap won’t wash off of the skin, and that one never gets the “squeaky clean” feel indicating when you’re really clean.
Water softener vendors are quick to point out both the squeaky and the slimy are illusions.
Here I’m going to borrow from an article written to sell softeners. Keep in mind that the source is not a peer-reviewed study from M.I.T. but a blog posting from a company that sells water softeners. I’m excerpting.
The Reality of Bathing in Hard Water
Have you ever toweled off after a shower, ran [sic] your fingers across your forearm or leg and felt a bit of squeaky friction? The concept of “squeaky clean” may have caused you to assume this meant all the greasy grime that was on your body had been washed away, and now your skin is sparkling.
Unfortunately, nothing could be further from the truth. But, perhaps it’s not your soap’s fault. Have you ever considered your water quality?
The short explanation is this … the squeaky clean feeling on your skin after a shower actually comes from soap that hard water was unable to wash away. Most bathing products don’t lather or clean well in hard water so soap residue gets left behind on your skin.
Imagine the soap scum you notice building up in your tub or that film that shows up on glass shower doors in need of a good cleaning. That’s what’s stuck to your body.
Squeaky clean skin is a straight-up lie. In fact, it means the exact opposite of what you’ve been led to believe. Your skin isn’t squeaky … it’s sticky. You’re not getting clean because the soap isn’t washing away, just like the soap scum on your tub. And, because it’s still on your body, you may get dry, itchy, flaky skin.
You see, the minerals calcium and magnesium are what make water hard. These hard minerals combine with soap to form what’s often called “curd,” which is just as gross as it sounds. The soap curd sticks to your skin and can clog up your pores and cause irritation. That sticky curd can also lead to brittle, unhealthy hair.
Soft Water: Slimy, Slick, or Silky Skin?
People who’ve recently installed a water softener in their home may notice their skin feels different after showering. Some describe it as a slippery feeling while others say their skin feels silky smooth after bathing in softened water.
Sometimes people complain about this sensation because they assume what they’re feeling is bath products that are left behind. Once again … this is the opposite of the truth.
As we’ve already explained, soap scum makes your skin sticky and dry. What you’re actually feeling after washing off with soft water is your body’s natural oils, which it uses to protect and moisturize your skin.
People who are unaccustomed to soft water say that they rinse and rinse, but the slick feeling won’t go away! That’s because it’s not soap product. It’s the way your skin is supposed to feel.
And now, you’ve been enlightened …
Pure Water Gazette’s Conclusion: There are lots of ways to look at things. Since the main purpose of bathing in softened water seems to be to get soap off of the body, a logical conclusion might be to stop using soap.
|
Places to visit on our websites |
Thanks for reading and be sure to check out the next Occasional!
|
|
|
|  | |