Pure Water Occasional, May 28, 2019 |
Top news stories center on floods. Lots of floods.
An extensive study by Environmental Science and Technology reveals long-term changes in temperature and precipitation are increasing the nitrogen content of freshwater supplies.
A 2019 consumer opinion survey conducted by the Water Quality Association found that Americans are far more interested in brands promoting a 100% Guarantee and a strong warranty than claims of being made or assembled in America. An informal opinion survey conducted by the Pure Water Occasional revealed that Americans are 38.6% more likely to buy a product advertised with a picture of a child drinking water than a product with a picture of baby ducks swimming in a bathtub.
Electric water bikes are being introduced this summer to ease commuter traffic in the San Francisco Bay area. More.
A major water main break in New Orleans closed schools, flooded streets, and caused a boil water alert.
A sobering 1,500 page United Nations report compiled by hundreds of international experts was issued in preliminary form. It reveals that humans are transforming Earth’s natural landscapes so dramatically that as many as one million plant and animal species are now at risk of extinction. The report is an exhaustive look at the decline in biodiversity across the globe and the dangers it creates for human civilization. More information.
Here's an old poster to remind you that National Garden Hose Day is no longer celebrated in August. The new date for National Garden Hose Day is June 18, right around the corner. Watch the Occasional for details.
California's new governor, Gavin Newsom, said in his State of the State address that at least one million Californians are being exposed to contaminated drinking water. He called the situation "a moral disgrace and a medical emergency," and called for a tax on drinking water to help disadvantaged communities clean up contaminated water. During his first week in office Newsom took his cabinet on a road trip to meet with Central Valley residents who for years have had to rely on bottled water for drinking and cooking.
Lake Dunlap
Heavy rains in Texas and the failure of a spill gate caused Lake Dunlap, on the Guadalupe River, to "dewater" completely. Downstream there was high water but no fatalities. Details.
Pequannock, NJ had to issue lead warnings to residents after city officials changed their testing method to include samples from older homes. 2016 testing recorded 3 parts per billion lead, but with older homes included in the sampling levels rose to 29 parts per billion, double the EPA allowable.
Mississippi River flooding
Spring floods on the Mississippi in 2019 "are among a new generation of Mississippi River crests that bear only passing resemblance to the floods of distant past." An excellent article on the realities of life along the Mississippi in the era of climate change in Scientific American.
The EPA is proposing a regulatory limit of 56 ppb for perchlorate, the rocket fuel. This is more than triple the 15 ppb limit proposed by the Obama EPA. The regulatory standard for perchlorate has been under consideration for over ten years, and the EPA has not actually issued a new regulatory standard for anything for over two decades.
Owners of as 500-foot replica of Noah's Ark have filed suit against their insurance company for failure to compensate them for $1,000,000 in damage to their property caused by rain. Details.
|
The “rotten egg” odor that people complain about in well water can come from many sources, but it is most commonly caused by “sulfur reducing” bacteria that give off a foul-smelling gas. The bacteria themselves are harmless–they don’t cause disease–but the gas they produce can cause horrible odors and smelly black staining in pipes and appliances.
In some parts of the country, most notably Florida, where hydrogen sulfide is common and very severe, the standard treatment is to spray the water into an open air tank, allow the noxious gas to escape into the atmosphere, then use a secondary pump to send the water from the tank into the home. Tanks of this type are expensive, need lots of space, and are subject to freezing in cooler climates.
Therefore, another type of treatment known as “precipitation/filtration” is preferred in most areas.
With this method, an “oxidizer” causes the trapped hydrogen sulfide gas to “precipitate” to elemental sulfur, then the sulfur is trapped in a filter. It’s a two-step process. The filter is most often carbon. Filter carbon, especially a specialty carbon called “catalytic carbon,” can perform both steps–precipitation and filtration–but unless the amount of H2S (hydrogen sulfide) is small, the carbon wears out quickly and has to be replaced. However, when the carbon is helped by a more powerful “oxidizer,” the carbon can last a very long time and the process can be very successful. Many “oxidizers” can cause the precipitation of the gas: air, chlorine, hydrogen peroxide, potassium permanganate, ozone, and more. For residential users, the most practical and the most easily maintained are aeration (air) and chlorination.
A full treatment system with chlorine looks like this–
1. A dry pellet chlorinator — a device that drops chlorine pellets into the well itself– followed by a carbon filter, or
2. A chemical feed pump, installed before the pressure tank, that feeds liquid chlorine (household bleach) into the water line. After the pressure tank, you must have a retention tank–usually 80 to 120 gallons–to give the chlorine time to work. After the retention tank, a carbon filter.
A full treatment system with aeration looks like this —
1. An “Aer-Max” system, which consists of a 10″ X 54″ treatment tank that is fed by a small air compressor. It is installed after the pressure tank, and it is followed by a carbon filter, or
2. A “single tank aerator” installed after the pressure tank. It is a backwashing filter with a special control valve that draws in air to “oxidize” the H2S so that it can be removed by the filter carbon in the bottom 2/3 of the tank.
Here are page links that show the various strategies. Many have installation diagrams.
The carbon filter used in any of these system (other than the single tank aerator) can be either a “backwashing” tank-style filter or a carbon block filter. If iron is present in the water, a backwashing filter must be used because a carbon block filter would be clogged quickly with iron.
Catalytic carbon is the carbon of choice with hydrogen sulfide, but any good carbon filter will work after proper oxidation.
Here are some places on our website to look for carbon filters —
Often the hard part of designing these filters is choosing and sizing the carbon filters. Do not hesitate to call or email us for help.
|
Getting Drinking Water From The Sea
by B. Sharper
|
Pure Water Gazette numerical wizard Bee Sharper rolls out some numbers on production of fresh water by sea water desalination.
Approximate number of desalination plants in the world as of 2013 — 17,200.
Daily production capacity of these plants in gallons — 23 billion.
Percentage of these plants that make potable water from sea water — 59%.
Percentage that make potable water from brackish water — 22%.
Percentage that make potable water from river water and wastewater — 9%.
Total dissolved solids (TDS) count of the saltiest of sea waters (e. g,.the Arabian Gulf) — 50,000 mg/L.
Total dissolved solids (TDS) count of most ocean water — 35,000 mg/L.
Typical chloride content of sea water — 19,000 mg/L.
Typical calcium content of sea water — 400 mg/L.
Typical sodium content of sea water — 10,500 mg/L.
Typical number of viruses present in one drop of sea water — 1,000,000.
Nominal pore size of a reverse osmosis membrane used for desalination — 0.0001 to 0.001 microns.
Nominal pore size of a nanofiltration membrane — 0.001 microns.
Psi equivalent of one bar of pressure — 14.5.
Pressure required to treat sea water by reverse osmosis desalination — 55 to 70 bars (800 psi to 1000 psi).
Pressure required to treat brackish water by reverse osmosis desalination — 15 to 35 bars (220 psi to 500 psi).
Percentage of salts, organics, and microbes that are removed from sea water by high pressure reverse osmosis — 99%+.
Estimated per gallon cost of producing fresh water by desalination with reverse osmosis –1/2 cent.
Desalination plant at Al Khaluf in Oman
|
We don't normally think of the clothing we wear as a water pollutant. The following article shows that we live in a complicated world.
|
The Invisible Nightmare in Your Fleece
Washing a single polyester jacket can send 1,900 tiny synthetic micro-fibers into waterways, where they can soak up toxins and get eaten by fish. So what is the outdoor industry doing about it?
by Mary Catherine O’Connor
|
|
Gregg Treinish is dismayed about what is coming out of his washing machine.
“What I’m seeing is shocking. Every couple of weeks, I clean out the filter and put the contents in a 32-ounce Ball jar,” says the founder of Adventurers and Scientists for Conservation (ASC), a nonprofit that trains outdoor enthusiasts to collect data for environmental researchers. After roughly two months, Treinish says, “the bottle is more than half-full of the crap that would have otherwise been shed right into the waterway.”
That crap is thousands of synthetic fibers shed from Treinish’s clothing during wash cycles (he captures them in an aftermarket filter), and the waterway is Montana’s Gallatin River. Treinish, whose organization receives financial support from a number of outdoor-gear companies, recently launched a campaign to track the flow of those fibers into fresh water. He plans to share that data with his funders.
What’s so bad about a few plastic threads? In 2011, British ecologist Mark Anthony Browne published a study describing the discovery of micron-scale synthetic fibers, mostly polyester and acrylic, in sediments along beaches the world over, with the highest concentrations appearing near wastewater-disposal sites. That strongly suggested that the micro-fibers came from apparel, a hunch he checked by filtering 1,900 fibers found in the waste-water from washing a single fleece jacket. A similar study at VU University Amsterdam in 2012 estimated that laundry wastewater is sending around two billion synthetic microfibers per second into Europe’s waters.
Of course, wool and cotton clothing sheds fibers, too. But those materials biodegrade. Plastics contain potentially harmful additives and can absorb toxins, including polychlorinated biphenyls (PCBs), that they encounter floating in waterways—and then get ingested by small organisms, crustaceans, and fish. These particles can accumulate in the animals’ guts and tissues, potentially weakening immunity or disrupting their endocrine systems. Less is known about how that payload may accumulate up the food chain.
New research shows that microfibers are even more abundant in our lakes and rivers than microbeads from shampoo and body wash, which have been banned in seven states. “We tested effluent from wastewater-treatment plants and found that 85 percent of the plastic it contained was fibers, whereas beads and other fragments only made up 13 percent,” says Sherri Mason, a professor of chemistry at the State University of New York at Fredonia. “Is it rocket science to assume those fibers are going to end up in some body of water?”
So while Treinish’s half-full Ball jar might not seem like a big deal, it represents a very thin slice of an incalculably large pie. And he believes that manufacturers are going to have to address their role in microfiber pollution sooner rather than later. “Apparel companies are realizing that once news about microfibers hits the mainstream media,” Treinish says, “they’re going to have some major issues to contend with.”
In 2013, Browne tried to form a coalition with big outdoor-industry brands, including Patagonia and Polartec, to track micro-fibers to their manufacturers. Most of the companies that responded declined to join Browne’s effort, saying they wanted to learn more about the problem—and how serious their role in it is.
That’s a hard question to answer. According to 2010 figures from the United Nations’ Food and Agriculture Organization, roughly half of all the clothes bought in developed countries—and 68 percent in the developing world—are synthetic. Your home is likely festooned with synthetic fibers as well, from carpeting to couch covers. But how often do you wash those items, compared with your polyester base layers, ski socks, fleece jackets, and pullovers?
Much of the clothing in question isn’t gear at all—hello, jeggings! But outdoor brands have long relied on the performance attributes of synthetic fabrics. Combined with chemical coatings and membranes, they can be both waterproof and breathable. While warm, odor-resistant wool has recently seen a bump in use for outdoor apparel, the market’s dependence on synthetics hasn’t changed.
As one industry source pointed out to me, so far there is no miracle fiber that is both high performing and environmentally benign. Conventionally grown cotton, for instance, requires significantly more water to produce than synthetics, utilizes far more land, and relies heavily on fertilizers and pesticides. And some manufacturers, environmentalists, and scientists, including Browne, suggest that the appliance industry should bear some of the responsibility, since washing machines don’t filter out small particles and add-on filters like the ones Treinish uses, which are designed to keep lint out of septic tanks, still don’t catch the tiniest fibers.
That said, the performance-apparel community is beginning to look inward.
The Outdoor Industry Association’s Sustainability Working Group, which represents 250 companies, has started to examine the issue, with cooperation from marine-debris specialists at the Ocean Conservancy. The investigation is still in its infancy, cautions Beth Jensen, OIA’s director of corporate responsibility. “You can’t mobilize an industry around an issue until you have all the facts,” she says. “But we do recognize the urgency around microfibers, and we want to move forward.”
Among individual brands, Patagonia has taken the most concrete steps in that direction. Though it declined Browne’s request to work together, the company is collaborating with the Bren School of Environmental Science and Management at the University of California at Santa Barbara to identify which synthetic materials in its supply chain shed fibers. “Everything we learn from this project will be useful information,” says Adam Fetcher, Patagonia’s communications director, “whether we find that we are part of the problem or not part of the problem or somewhere in between.”
Other brands I called, like Polartec and the North Face, elected not to comment. Columbia Sportswear, which was the target of an online petition initiated by anti-plastics group the Story of Stuff, provided a statement from Peter Haney, its manager of corporate responsibility. “Columbia is in the early stages of reviewing Dr. Browne’s initial research,” he wrote. “We will continue to be involved through industry-wide collaborations like OIA’s Sustainability Working Group.”
For now, the most significant progress is taking place in Europe, where a new research consortium called Mermaids, funded by the European Commission and promoted by the Netherlands’ Plastic Soup Foundation, is dedicated to reducing microfiber shedding by 70 percent. The first step is to identify the worst culprits. Browne and others are working to develop a process that can trace a polyester or nylon thread back to its point of origin by identifying the fingerprint left behind by manufacturing dyes and chemicals. The plastics in medical implants go through a battery of certifications before they’re used in human bodies, he argues. Why shouldn’t consumer plastics undergo equally rigorous trials to determine their impact on aquatic ecosystems?
That may be a long way off. “Hey, I live in Maine, and I could not live without my fleece,” says Kara Lavender Law, a microplastics researcher at the Sea Education Association. “The big question is: Where should we be using plastic? It’s clearly a very useful, beneficial material, but maybe it’s time to reconsider wool or other materials that were replaced with synthetics.”
|
"Flushable" Wipes Clog NY Sewers |
New York city’s 14 wastewater treatment plants have to process 1.3 billion gallons of wastewater per day (twice that when it rains) gathered from 6,000 miles of pipes. The largest of these, the Newtown Creek Plant, which covers about 50 acres, processes about 310 million gallons daily. The plant spends around $19 million per year just cleaning out clogs caused mainly by so-called “flushable”wipes. When these wipes congeal with grease also dumped down drains, “fatbergs” result.
Efforts at legislation and judicial action to keep the wipes from being sent down the city’s toilets have largely failed due to industry resistance.
|
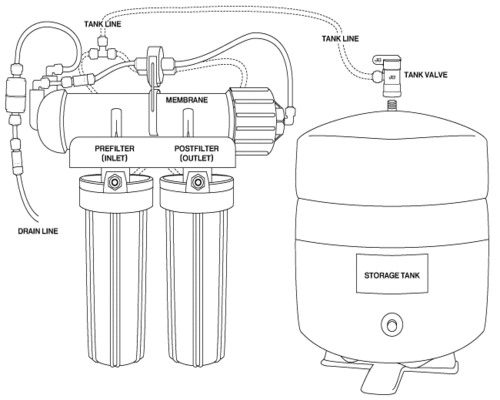
How Reverse Osmosis Shutoff Systems Work
|
Modern undersink reverse osmosis units use a simple but effective shutoff device to turn off water production when the unit’s storage tank is full. The shutoff system monitors the pressure in the storage tank and shuts off water coming into the RO membrane when tank pressure reaches approximately 2/3 of the pressure of the incoming tap water.
Thus, a “full” tank in a standard RO unit is defined by the pressure of the water entering the RO unit. If incoming pressure is 60 psi, a full tank holds about 40 psi; if incoming pressure is 50, a “full” tank holds about 30 psi. Shutoff is done at around 2/3 of incoming pressure because as the tank fills increasing back pressure makes production less efficient. It isn’t practical to fill the tank past 2/3 full.
The Payne brand shutoff pictured above is installed as follows:
1. After water leaves the RO unit’s prefilter, it enters the “In” port of the shutoff valve, lower right in the picture. It then makes a horseshoe turn and exits the “Out” port, lower left in the picture, through which it flows to the inlet side of the RO membrane.
2. When the “permeate” water (the product water of the RO unit) leaves the other end of the membrane housing, it flows to one of the “tank” ports on the other side of the shutoff valve. It doesn’t matter which port it enters, since the “tank” ports are interchangeable and water flows either way on the permeate side of the valve. Water then makes a horseshoe turn inside the top side of the valve and leaves through the other tank port. From there it flows to the storage tank.
3. The two halves of the valve are separated by a piston, which keeps the permeate water on one side and the incoming tap water on the other. As long as the pressure on the tank side is less than 2/3 the pressure on the tap water side, the piston remains open and the unit continues to produce water. As the RO produces water and slowly fills the storage tank, however, pressure on the tank side of the piston eventually becomes strong enough to force the piston toward the tap water side and shut off the incoming tap water, stopping production. The RO unit stays off until enough water is removed from the storage tank to drop the pressure on the tank side of the piston, allowing tap water pressure to push the piston toward the tank side and start RO production again.

The Flowmatic shutoff valve above works exactly like the Payne valve, although the flow pattern is straight through rather than horseshoe style. In other words. water enters lower right and flows straight ahead through the valve and out the other side.
It is important to know that in order for the shutoff system to work, a check valve (one-way valve) must be installed in the permeate tube between the membrane and the shutoff valve. Without the check valve to hold the back pressure from the tank, the shutoff valve cannot function.
The Payne shutoff valve is clipped to the membrane housing of the unit above. The tubes on the right side carry tap water to the membrane. On the left, or “tank” side, permeate water leaves the membrane, passes through the cigar-shaped check valve, and enters one of the shutoff valve’s “tank” ports. It leaves via the other tank port and flows to the tee, which sends it to the storage tank.
Note that the inline check valve will not be found on most RO units, since RO manufacturers usually prefer a tiny, inexpensive check valve that”s contained in the elbow fitting where the permeate water leaves the membrane housing.
Troubleshooting your shutoff system
If you hear water running to drain more than you think it should or have some other reason to suspect that your RO unit isn’t shutting off properly, here’s an easy test you can do to check its performance.
Run a few glassfuls of water from the RO unit to start the unit producing water, then turn off the RO faucet and remove the drain line from the drain saddle connected to the undersink drain pipe. Drop the end of the drain line into a bottle or pan so that the drain water trickles into the container. Next, turn off the valve at the top of the RO storage tank. Water should stop flowing from the drain line within a couple of minutes. When it stops, leave the valve off, empty the container, put the tube back into the container, and come back in 10 minutes. If there is no water in the container, the shutoff system is working perfectly. The unit is shutting off and holding its shutoff.
If the drain fails to shut off, you need to find the reason. The main suspects are the shutoff valve or the check valve, not necessarily in that order.
If the drain shuts off initially, but comes back on during the 10 minutes, continue to watch it. If it comes on, runs briefly, then shuts off, and repeats this pattern over and over, you need a new check valve.
|
A Common Sense Approach to Residential PFAS Treatment
by Emily McBroom and Gene Franks
|
Most of the information now available about how to remove PFAS from water focuses on the gigantic carbon filters made to treat the millions of gallons of water per day required by cities. Also mentioned are the very small residential drinking water filters, mainly carbon filters, that have received NSF certification for PFAS removal from drinking water.There is a lot of confusion about the residential applications that fall between the gigantic and the tiny.
Here are some things to consider about residential applications for PFAS reduction.
1. Consensus is that three treatment strategies work with PFAS: carbon filtration, reverse osmosis, and ion exchange. Of these, carbon (GAC) filtration seems most practical for municipalities. GAC and reverse osmosis both work well for residential users, with reverse osmosis an easy first choice for drinking water. One agency tested eleven separate undersink reverse osmosis units and found that they all removed PFAS well. Several pretty unassuming carbon filters have gained NSF certification for PFAS reduction.
2. Those who recommend treatment equipment for residential applications almost always make an unfounded assumption that homes must use point of entry equipment, treating all the water going into the home. We find no convincing information to indicate that PFAS in water is anything other than an ingestion issue. According to the CDC’s Agency for Toxic Substances Disease Registry: “Studies have shown that only a small amount of PFAS can get into your body through your skin. Therefore, showering and bathing in water containing PFAS should not increase exposure. Washing dishes in water containing PFAS should not increase exposure.”
3. For point of entry treatment, we found that almost all recommended treatments are simply scaled-down versions of the strategies developed for municipalities. Usually there is no rationale stated that would justify the sizing recommendation. Recommended sizes range from extra large to even larger. We found EBCT (Empty Bed Contact Time) recommendations for PFAS ranging from 6 to 16. Ten is a common recommendation. Using an EBCT of ten, to provide a modest service flow of five gallons per minute for a residential whole house filter for PFAS one would need almost 7 cubic feet of granular carbon. That’s a 21″ X 72″ carbon tank, or three or four 12″ X 52″ tanks installed in series, or five 10″ X 54″ tanks installed in series. One authority recommends “at least 200 pounds of GAC” for residential whole house treatment–a 7 cubic-foot filter array. That’s a lot of equipment to assure PFAS-free water for flushing toilets. The most commonly suggested point of entry system is for two 12″ X 52″ GAC filters installed in series, without regard to family size. This would provide an EBCT of 6 at 5 gpm.
4. No one offers information about PFAS performance for whole house sized carbon block filters, although carbon block units might offer the most practical PFAS whole house option.
The Obvious Conclusions
Our advice to consumers is get an undersink RO unit. If you don’t want reverse osmosis, get a high quality undersink or countertop carbon drinking water filter with an ample amount of carbon, and service it regularly. Ignore PFAS as a point of entry treatment issue, but don’t drink water from the bathtub. Treat your drinking water well, and add a whole house carbon filter if you want to, but you don’t have to get a box-car sized filter that competes with the city water department because if your drinking water is taken care of, a little PFAS in the shower water won’t matter.
|
Places to visit for additional information:
|
Thanks for reading and be sure to check out the next Occasional!
|
|
|
|  | |